Bonesupport, a Swedish medical tech firm, specializes in CERAMENT, the only FDA-approved antibiotic-eluting bone filler available. When surgeons need to address bone defects and fight infection simultaneously, Bonesupport's product is often the only option. Headquartered in Lund, it trades on OMX Stockholm.
CERAMENT is a synthetic bone graft that can be loaded with antibiotics before surgery. It offers structural support, releases antibiotics locally as it erodes, and is gradually absorbed to allow natural bone growth. The company's revenue mainly comes from a U.S. distributor network, making up about 84%, with the rest from Europe.
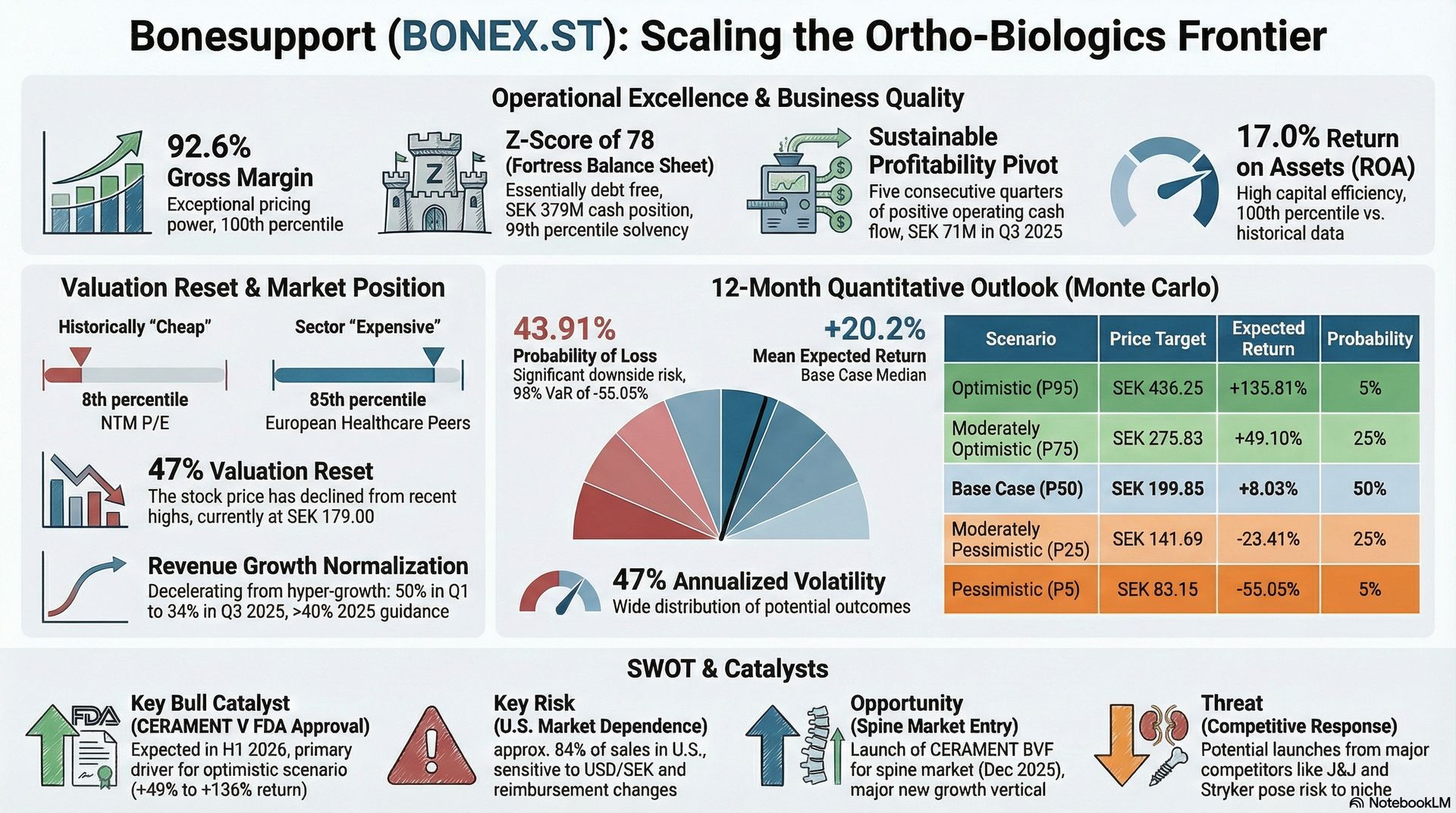
Having shifted from a loss-making development-stage company to a consistently profitable business—marked by five straight quarters of positive cash flow—Bonesupport's stock has nonetheless fallen around 47% from its peak. It now trades at its 8th percentile on forward P/E compared to its five-year history. This suggests either the market discerns something not evident in the financials or an investment opportunity has appeared.
Bonesupport displays a unique profile: operational excellence paired with valuation skepticism. The company has achieved what many in medical technology struggle with; a sustainable shift from development-stage losses to profitable growth, backed by impressive margins and a true regulatory advantage. Despite this, the market values the stock as if this success is fragile or fully priced in.
The core question isn't whether Bonesupport is a solid business. Instead, it's whether a 47% drop offers an entry point that justifies the risks of execution, or if it simply reflects the market's realistic view of a company moving from rapid expansion to steady growth.
The Fundamentals
The company's key indicators are exceptionally strong. Bonesupport reports a gross margin of 92.6%, placing it at the 100th percentile within its historical range. This is outstanding not only for a medical device firm but by any standard. EBITDA margin has risen to 21.9%, with operating margin climbing to 27%, both reaching their historical highs. The company carries virtually no debt—its total debt-to-equity ratio is only 1.35%, and it holds SEK 379 million in cash. The Altman Z-Score of 78 ranks in the 99th percentile within its sector, signaling robust financial stability.
Return metrics have improved along with margins. Return on assets is at 17.0%, and return on total capital is 19.9%, both at their peak. Return on equity is 20.3%—the 67th percentile—lower mainly because the company uses modest leverage, which reduces the amplification effect. Free cash flow yield has reached 1.75%, which, though modest in absolute terms, is in the 90th percentile historically for a company that previously consumed cash rather than generated it.
Revenue growth, despite slowing down, remains strong. Constant exchange rate growth slowed from 50% in Q1 2025 to 34% in Q3, but management maintains its guidance of over 40% CER growth for the full year. The cumulative growth from Q1 to Q3 is 44%, supporting this target. US sales in Q3 were around USD 19.2 million, and the company expanded its surgeon training programs and hospital system listings successfully.
The valuation landscape highlights a key tension. Compared to its own history, Bonesupport appears very cheap: forward P/E at the 8th percentile, price-to-sales at the 5th percentile, and EV/EBITDA at the 7th percentile. However, relative to European healthcare peers, the picture flips: P/E is at the 85th percentile, and EV/EBITDA at the 92nd percentile. The market seems to suggest that Bonesupport's growth premium has diminished compared to peers, but the discount relative to its historical valuation indicates that investor perception has shifted specifically regarding this stock.
Between the Lines
The market implies, through its pricing, that Bonesupport's strong operational results are unsustainable or that hidden risks exist. However, the data paint a different picture: a company performing at record-high levels across nearly every operational metric, backed by a unique regulatory advantage and a growing market opportunity.
Three main factors explain this disconnect. First, concerns over slowing growth. The shift from 50% quarterly growth to 34% has unsettled investors used to rapid expansion. Yet, 34% growth in constant currency would be impressive for most healthcare firms; the key issue is the growth trajectory's impact on investor sentiment. Second, regulatory uncertainties. The approval of CERAMENT V, an advanced product with improved antibiotic delivery, by the FDA, expected in H1 2026, is crucial. Any rejection or delay could significantly alter the growth outlook. Third, currency fluctuations. With 84% of revenues in US dollars but costs and reporting in Swedish kronor, SEK/USD exchange rate changes cause notable earnings translation effects.
Investors must decide if a 47% drop sufficiently offsets these risks or if the market has overreacted. Bonesupport's competitive advantages remain strong. Its 92% gross margin provides substantial operating leverage as it grows. The shift to profitability seems structural rather than temporary. However, significant US market reliance (84% of sales), dependency on distributors (around 34% commission structures), and regulatory uncertainties suggest the valuation decline might be justified.
Risk Modeling
Our model incorporates a 47% annualized volatility estimate based on recent price movements and equilibrium drift assumptions. The results suggest a distribution with notable upside potential but also significant downside risk.
The likelihood of loss is 43.9%, meaning just over four in ten simulated paths fall below the current price. The 95th percentile Value at Risk indicates a maximum expected loss of 55.1% across 19 out of 20 scenarios, while Conditional VaR (the average loss in the worst 5%) reaches 64.3%. The average maximum drawdown across all paths is 38.9%, implying that investors should anticipate considerable volatility even in paths that ultimately turn out profitable.
Risk-adjusted metrics show a mixed outlook. The Sharpe ratio of 0.27 is below the generally accepted threshold of 0.5, suggesting that expected returns may not adequately compensate for volatility. Conversely, the Sortino ratio of 0.91 (which accounts only for downside volatility) and the Omega ratio of 2.56 (which compares probabilities of gains versus losses) indicate that return asymmetry could benefit patient investors. The gain/loss ratio of 2.0 demonstrates that when gains occur, they tend to be twice as large as losses.
Model limitations: The simulation assumes equilibrium drift and cannot capture binary regulatory events like FDA approval or rejection. While currency effects are included in the historical volatility, they may not accurately predict future SEK/USD movements.
The Upside
Various specific triggers could narrow the valuation gap. The most notable is the anticipated FDA approval of CERAMENT V in H1 2026. This improved antibiotic-eluting product would grow the target market and strengthen Bonesupport's regulatory advantage. A smooth approval could drive significant multiple expansion as investors incorporate an extended growth trajectory.
Additionally, the launch of CERAMENT BVF for the spine, starting in December 2025, could serve as another catalyst. The spine market is considerably larger than the trauma and arthroplasty segments where Bonesupport currently operates, and initial surgeon feedback and hospital listing data from Q1 2026 will be closely monitored. Strong initial adoption could prove that the company's platform effectively applies to orthopaedic uses.
Moreover, clarity on reimbursement could boost the stock. The current NTAP offers premium reimbursement but has an expiration risk. Achieving permanent DRG codes would eliminate this uncertainty and improve visibility into long-term pricing. Lastly, the company’s Capital Markets Day scheduled for Spring 2026 may share strategic plans regarding its significant cash reserves and update guiding growth expectations, either confirming or adjusting previous forecasts.
The Downside
The primary risk is regulatory disappointment. The FDA approval process for CERAMENT V is a binary event with significant consequences. While approval is anticipated, receiving a Complete Response Letter or facing outright rejection could severely impact the company's short-term growth and damage market confidence in its regulatory abilities. Investors should watch FDA communications carefully; any requests for additional data or delays should be seen as early warning signals.
Regional concentration presents a structural vulnerability. With 84% of revenues coming from the United States, Bonesupport is heavily exposed to a single regulatory environment, reimbursement system, and competitive landscape. The European market has stagnated, especially in Germany and the UK, removing the natural diversification benefit. A year-over-year decrease in European revenue exceeding 10% would suggest regional weakness is worsening instead of stabilizing.
Currency fluctuations influence both reported earnings and competitive positioning. The SEK has varied significantly against the USD, causing translation effects that can hide or exaggerate actual operational performance. Investors should focus on constant-currency metrics, as headline growth figures may differ greatly from real business momentum depending on exchange rate changes.
Lastly, competitors with substantial resources, like Johnson & Johnson and Stryker, could respond aggressively. They have extensive distribution channels and R&D budgets to develop rival antibiotic-eluting products. While Bonesupport's regulatory head start and clinical evidence provide some protection, the orthopaedics market has a history of incumbents reacting strongly to new entrants.
DISCLAIMER
The information provided in this newsletter is for educational and informational purposes only and does not constitute investment advice, financial advice, trading advice, or any other type of advice. The content should not be interpreted as an offer, solicitation, or recommendation to buy, sell, or hold any financial instrument.
The author is not a licensed financial advisor, broker, or dealer and is not authorised or regulated by any financial supervisory authority. All investment decisions involve risk, including the potential loss of principal. Past performance is not indicative of future results. Readers should conduct their own research before making any investment decisions.